Back to: SENIOR TWO CHEMISTRY NEW CURRICULUM
pH AND THE STRENGTH OF ACIDS AND ALKALIS
Strength of acids and alkalis
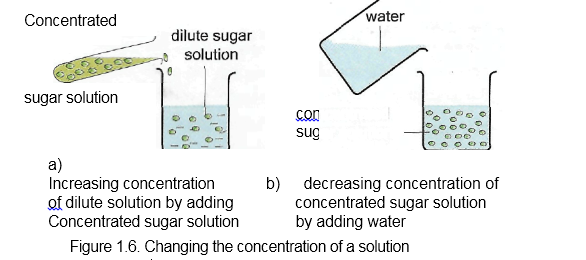
“The term concentration tells us how much a substance is dissolved in 1 dm3/litre of solution. The concentration of a solution can be changed. This can be by dilution (i.e., by adding water to the solution), this decreases the concentration. Or by adding more of the substance to the solution, thereby increasing the concentration as in Figure 1.6. However, the strength of an acid or alkalis cannot be changed.
The term strength refers to how easily an acid or alkali dissociates when dissolved in water. A strong acid, like hydrochloric acid, dissociates easily in water. A weak acid, like ethanoic acid, does not fully dissociate when dissolved in water. It has much lower concentration of hydrogen ions in its solution.
Since the strength of an acid or alkali cannot be changed, hydrochloric acid is a strong acid whether it is diluted or concentrated. Ethanoic acid remains a weak acid, whether it is diluted or concentrated. The strength of an acid or alkali can be shown by a pH scale
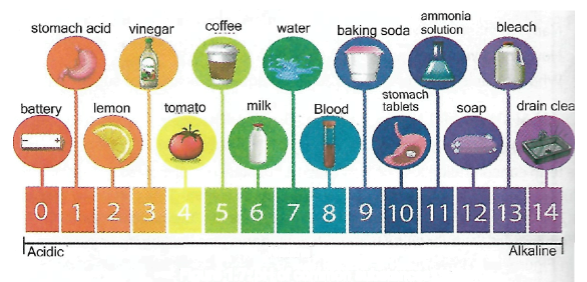
The phi scale is a set of numbers used to indicate whether a solution is acidic neutral or alkaline. For example, acids have pH values less than 7; alkalis have pH values greater than 7; and a neutral solution has a pH value of exactly 7. The pH of some common substances is as shown in Figure 1.7.
Acitivity
In this activity, you will work in groups and use a universal indicator papa” decide the strength of the substances you are given.
What you need
- carbonated water • coca cola
- diluted bleach • diluted liquid soap
lemon juice • tap water
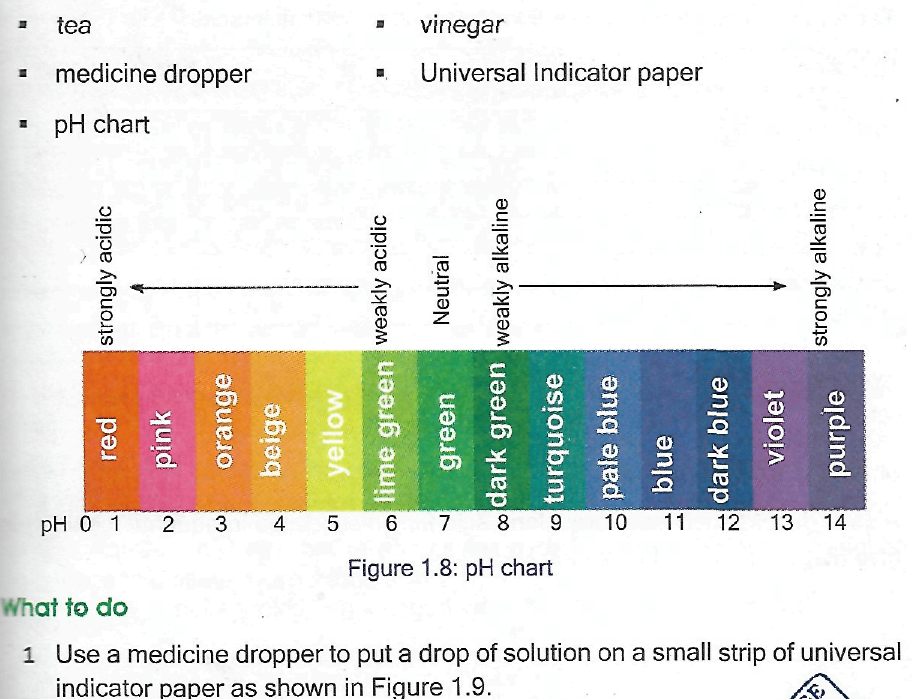



QUESTIONS
1 What does the pH scale measure? What is the pH of a neutral solution?
- When a liquid has a pH 0, what type of liquid is it?
- Which colours does universal indicator give in acids?
- Which colours does universal indicator give in alkalis?

THIS NOTES HELP ME ALOT